Multiplexed Diagnostics
Make informed decisions, faster
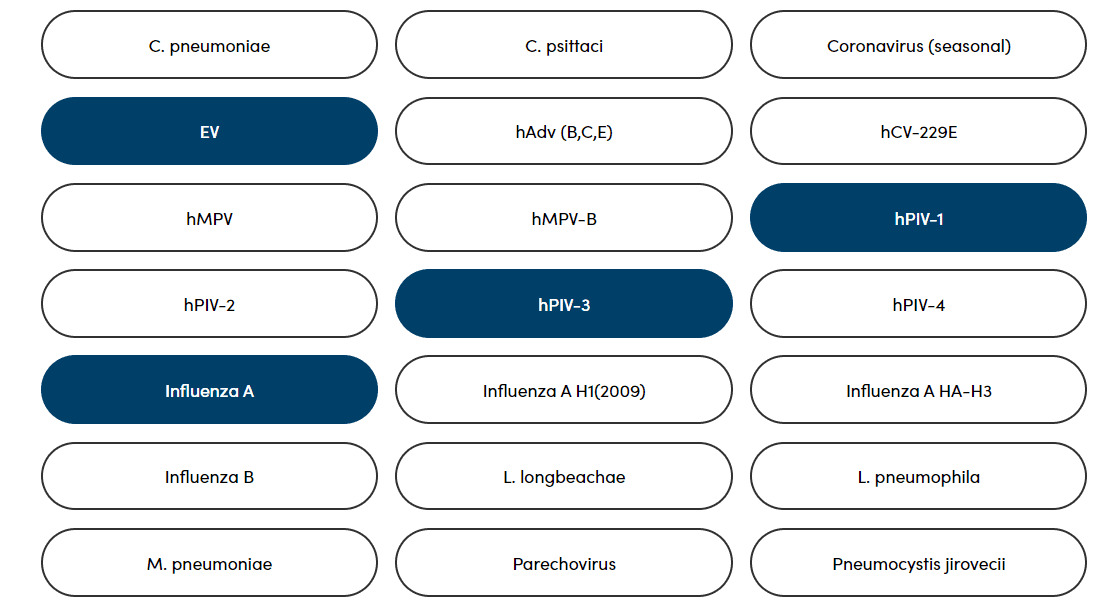
Combine testing for clinically-relevant pathogens, including bacteria, viruses, and parasites, plus antimicrobial resistance and genotyping.
ACKNOWLEDGEMENT OF COUNTRY
AusDiagnostics acknowledges the traditional custodians of the land on which our office stands and pays tribute to the indigenous generations of the Gadigal nation, respecting elders past, present and future.





